A Guide for Discussing Your Decentralized Trial Strategy with Your CRO

Eight Parameters to Achieve Site- and Patient-Centric Decentralized Trial Capabilities
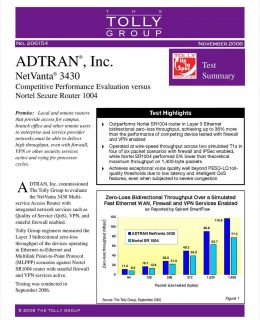
Disruptions preventing or limiting clinical trial site access to patients, monitors, and CRAs are propelling long-lasting changes in the research industry, supported by the adoption of trial virtualization technologies. The urgency to adapt to evolving requirements for both patients and sites is prompting sponsors to engage in new discussions with their CROs.
- Are you prepared to adopt a decentralization strategy?
- Can you respond to urgent remote requirements for patients and sites?
- Have you and your CRO evolved your technology approach to support fully remote study conduct?
- Will your strategy meet future standards for transforming trials?
This guide provides eight parameters to help sponsors optimize their path to virtualization across key site- and patient-centric capabilities.